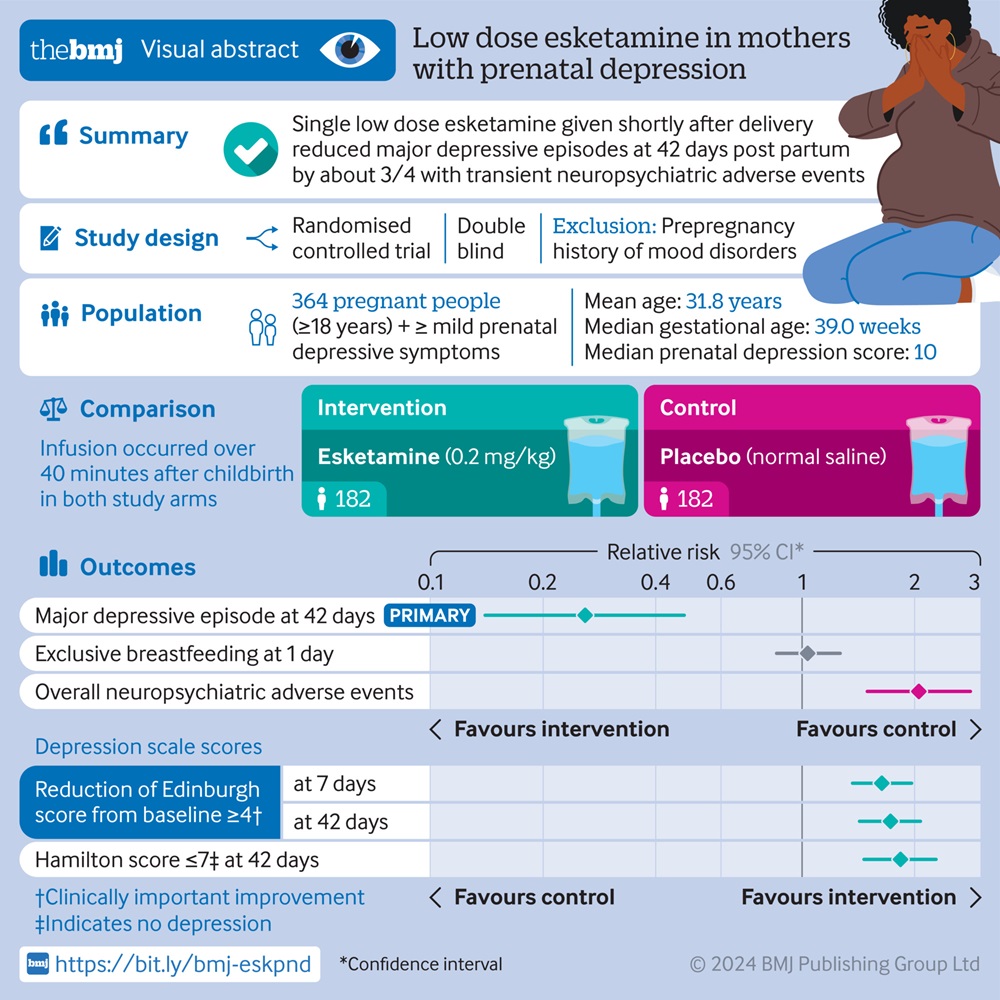
Esketamine Depression Trial Raises New Questions
If you follow new depression treatments, you have probably seen bold claims about fast relief and major progress. The problem is that trial results often look cleaner in headlines than they do in the actual data. That matters now because esketamine depression trial coverage can shape patient expectations, clinic demand, and how doctors frame a hard treatment choice. Some people with treatment-resistant depression do improve with esketamine. But the latest reporting also points to familiar pressure points, including dropout rates, study design, and the gap between a measured response and a lasting real-world benefit. So what should you pay attention to? Look past the sales pitch. The details are where the story lives.
What stands out right away
- The latest esketamine depression trial discussion centers on how much benefit patients actually gained, not just whether the drug beat placebo.
- Study dropout and trial design can change how strong the result looks on paper.
- Esketamine may help some patients with treatment-resistant depression, but it is not a simple fix.
- Readers should separate symptom score changes from durable, day-to-day recovery.
Why this esketamine depression trial matters
Esketamine, sold as Spravato, already has a foothold in psychiatry. It is approved for adults with treatment-resistant depression and for depressive symptoms in adults with major depressive disorder with acute suicidal ideation or behavior, under specific conditions. That gives every new study extra weight.
But approval is not the same as settled science. Psychiatry has a long history of treatments that looked strong in select trials, then produced messier results in regular practice. Think of it like a carefully plated restaurant meal versus what comes out when the same recipe hits a busy home kitchen. The ingredients matter, but so do the conditions.
Trial wins matter. Real-world staying power matters more.
What the latest report suggests about benefit
Based on MedPage Today’s coverage, the central issue is not whether esketamine shows any signal at all. It is how large that signal is, and how confidently you can interpret it once missing data, attrition, and trial structure enter the frame.
That is the part many readers miss. A treatment can produce a statistically detectable improvement while still leaving open a basic practical question: will enough patients feel better in a meaningful way to justify the cost, monitoring burden, and side effect profile?
That is the real test.
Esketamine requires supervised administration in a certified healthcare setting because of risks such as dissociation, sedation, and blood pressure increases. Patients also need post-dose monitoring. So even a modest benefit has to be weighed against a very real treatment load for both patients and clinics.
How to read the esketamine depression trial without getting fooled
1. Check the endpoint, not just the headline
Depression studies often lean on rating scale changes, such as shifts in symptom scores over a short window. Those measures matter, but they can also flatten the bigger picture. Did patients return to work? Sleep better? Stop cycling through crisis care? A score drop is useful. It is not the whole story.
2. Watch for dropout rates
If many participants leave a study early, interpretation gets shaky fast. Why? Because patients who stop treatment are often not random cases. Some leave due to side effects, lack of effect, logistics, or worsening symptoms. And if a trial has to estimate missing outcomes, confidence can thin out.
3. Ask who the treatment is really for
“Treatment-resistant depression” sounds precise, but it covers a mixed group. Some patients have failed several antidepressants. Others have more complex psychiatric or medical profiles. The narrower the trial population, the more careful you should be before applying the result to everyone sitting in a clinic waiting room.
4. Separate speed from durability
Esketamine gets attention because it may work faster than standard oral antidepressants for some patients. Fair point. But speed is only half the job. If the benefit fades, or if maintenance becomes too hard to sustain, the early gain may not hold up where it counts.
What patients and clinicians should ask next
Honestly, this is where the hype usually outruns the evidence. If you or someone you care about is considering esketamine, the smart questions are practical ones.
- How much improvement did patients see, in plain terms?
- How many people stopped treatment, and why?
- What side effects were common during supervised dosing?
- How long did benefit last after the initial response?
- What alternatives were compared, and were they strong comparators?
- How hard is the treatment schedule to maintain in normal life?
Those questions may sound obvious, but they cut through marketing language fast.
The bigger context around Spravato and treatment-resistant depression
Spravato entered a field hungry for faster options. That demand is real. Major depressive disorder can be disabling, and treatment-resistant depression leaves many patients stuck after multiple medication trials. In that setting, any therapy with a new mechanism will attract intense interest.
But interest can distort judgment. The ketamine family of treatments has often been framed as a dramatic break from standard antidepressants. Sometimes that framing is fair. Sometimes it turns a nuanced clinical tool into a symbol of hope that no single study can support.
And that is risky for patients.
A veteran observer of psychiatry coverage learns to watch for one thing above all else: does the article explain tradeoffs, or does it sell momentum? If a report skips the friction, you are not getting the full picture.
What this means if you cover healthcare or behavioral health
The media angle here matters too. Reporters and editors should avoid treating every positive signal as a seismic shift. Better coverage explains effect size, uncertainty, study population, and practical constraints. It names the source of enthusiasm, but it also tests it.
For publishers in healthcare, finance, regulation, or adjacent sectors, the lesson is simple. Readers do not need louder claims. They need clean framing. The strongest stories on psychiatric drugs tell people where the evidence is solid, where it gets thin, and what remains unsettled.
Where the evidence needs to go next
The next wave of esketamine depression trial reporting should focus less on novelty and more on patient-relevant outcomes. That means longer follow-up, cleaner handling of dropout, sharper subgroup analysis, and more transparent discussion of adverse events and treatment burden.
What would really move the field? Better evidence on who benefits most, who is likely to stop, and whether repeated supervised dosing produces durable gains outside tightly managed study conditions. Until then, the right stance is neither cynicism nor cheerleading. It is disciplined curiosity.
Psychiatry does need better tools. The question is whether the next esketamine headline reflects a true step forward, or just another polished result that gets softer the closer you look.